Dalent Medical is committed to creating better solutions for physicians to help restore health and well-being to patients living with sinus-related conditions. The company’s first FDA-listed product, the Sinusleeve™ Balloon Sinus Dilation Sleeve is used to treat chronic sinusitis, a condition that affects more than 30 million people annually, according to Center for Disease Control.

Shawn McCoy, CEO

Peter Flores, CTO / CBDO

Dr. Agustin Arrieta, CMO

Michael Dollar, VP Marketing & Commercialization
After operating one day, ear, nose, and throat doctor, Gus Arrieta, was struck with how many complicated and expensive medical devices he had used for just one procedure. Staring at the pile of packaging from the devices, he was inspired to create a simpler, better, and more efficient device for balloon sinus dilation — the Sinusleeve™ Balloon idea was born. The doctor teamed up with Peter Flores, a biotech engineer, and they began working on a prototype.
The working title for the project was “Dale ENT”, dale (dah-leh) meaning “go for it” in Spanish. The “let’s do it” mentality ended up becoming part of the company’s DNA and the project code name was the perfect fit being a perfect fit for the company brand. Dalent Medical added experienced medical device executive Shawn McCoy to the team, raised an investment round, and it was off to the races. In 2019, Dalent Medical was granted a patent for the Sinusleeve™ Balloon, registered as an FDA manufacturer, and began marketing and sales to ENTs.
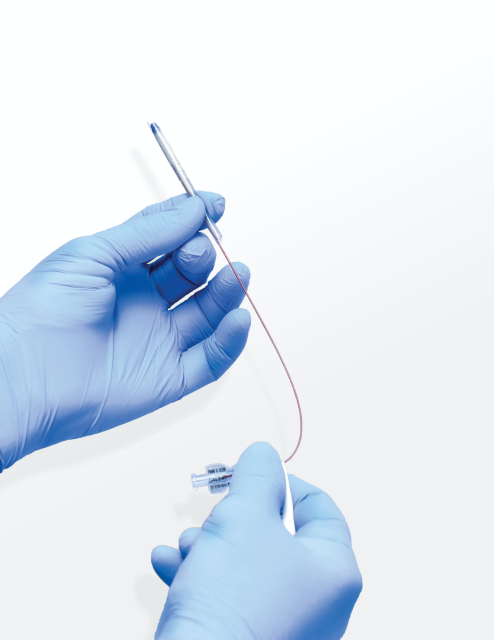


The Sinusleeve™ Balloon is a single-use, disposable, balloon sinus dilation (BSD) sleeve device used for treating chronic sinusitis. Developed and manufactured in the United States, the device fits over rigid, flexible and reusable positioning and suction devices by way of a conforming sleeve. The Sinusleeve™ Balloon accommodates instrumentation of varying sizes and curves, allowing access, simultaneous suction and balloon dilation all in one.
Chronic sinusitis is a condition which affects more than 10% of the population within the United States. The amount of surgeries to treat this condition continues to rise as less invasive techniques offer improved patient experience, measurable quality of life improvements, and lower healthcare costs. Dalent Medical is uniquely positioned to capitalize on this market by disrupting the category with a more innovative device developed by ENTs for ENTs.



Before the Sinusleeve™ Balloon, ENTs needed more devices on-hand to execute sinus dilation procedures. Now with a more versatile sleeve, surgeons can streamline costs without limiting their instrument options. In summary, this device is purpose built to enable doctors to provide better care and lower costs.
Following the launch of the Sinusleeve™ Balloon, Dalent Medical is continuously working on additional medical devices that align with their vision of providing patients and doctors the best tools with the highest value. Headquartered in Miami, the company also has an office in Little Rock, Arkansas.













